Why are early-onset colorectal cancer rates spiking?
A story of how your microbiome breaks your DNA, and what we can do about it
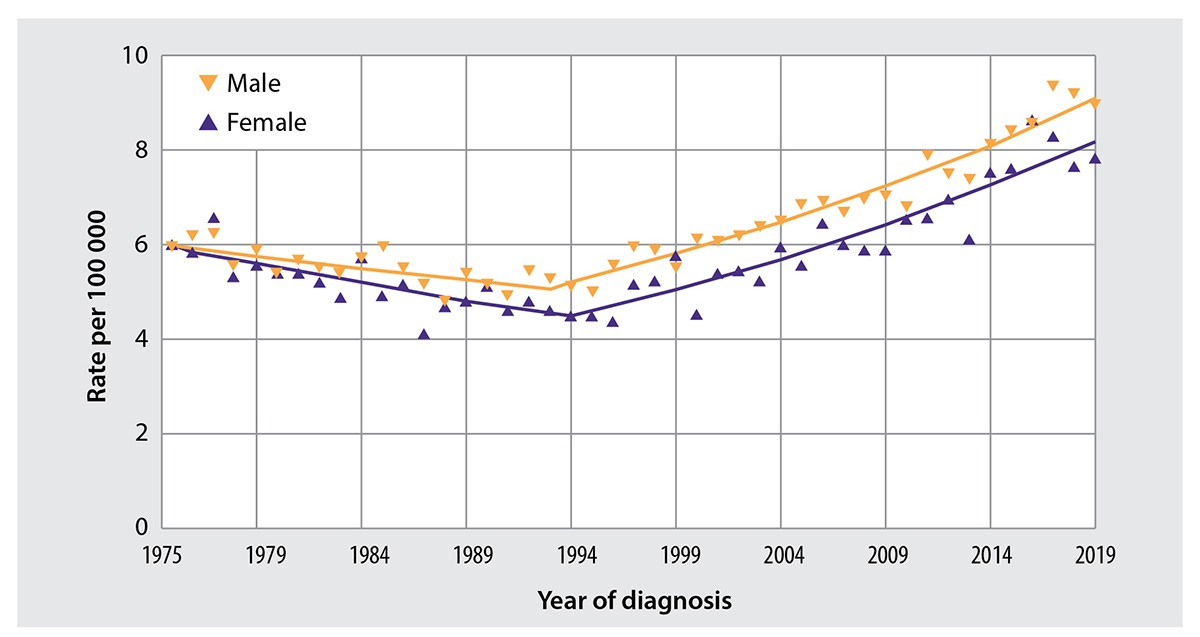
You may have heard that early onset colorectal cancer has doubled in the past 30 years. This claim is true, but undersells the phenomenon - “early onset” is medically defined as “under the age of 50.” If you focus only on colorectal cancer in children (individuals under 18 years of age), there has been a ~400% increase in incidence1.
Why? To some extent the answer to this question is unknowable2, but a wide range of explanations have been proposed, from obesity to microplastics to dietary changes. Many of these influence colorectal cancer risk by affecting the microbiome, shifting it away from an enrichment of symbiotic bacteria and towards a higher ratio of aggressive, pro-inflammatory microbes.
But how does changing the microbiome cause cancer? The answer, in many cases, is very direct.
Bacteria can break your DNA
The microbiome is not a static environment. It is an ecosystem containing hundreds of different bacterial species, many of which are directly competing with each other for resources. They are spread out throughout the entire GI tract, sitting above the epithelial cells that form your gut while being policed over by vigilant immune cells. In the same way that a forest ecosystem changes over time, the composition of your particular microbiome is never stagnant. The microbes are not only changing in proportion, but also in character - mutations enable some bacteria to be more efficient in scavenging resources, and others more capable at evading the immune system. Some bacteria have a more direct ability to dominate the microbiome niche: they create toxins that kill other bacteria and any immune cells that might try to stop them.
This brings us to pks+ E. coli. This particular strain of bacteria is among the most problematic, specifically because it creates a toxin that is designed to staple DNA strands together. Bear with me for a moment to discuss the biochemistry, because the mechanism is fascinating.
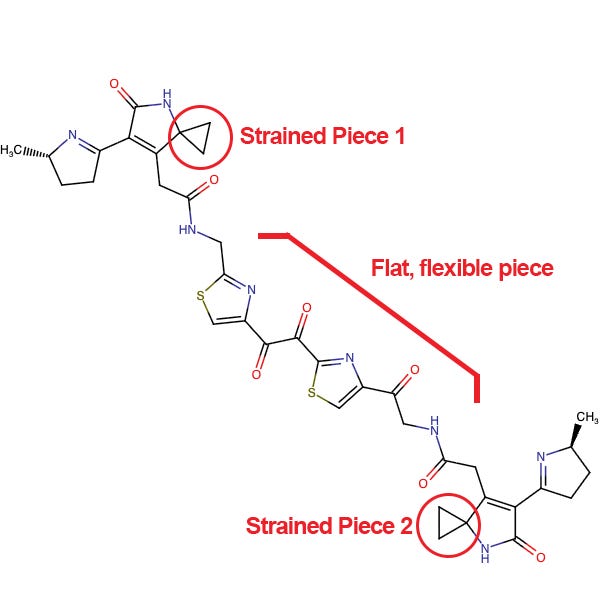
The toxin in question is colibactin. Below is the chemical structure - I’ve circled the most interesting bits:
It may not look like much. It’s a bit large and unwieldy compared to what most bacterially-derived toxins look like. But looking deeper, there are two key components:
On each end, there are chemical structures that are geometrically a bit similar to a nucleotide that you would see in DNA, except with extremely unstable and strained cyclopropane “warheads” (the triangular pieces).
In the middle, there’s a flexible linker with aromatic rings every few carbons. This looks a bit like a ladder, with two “steps.”
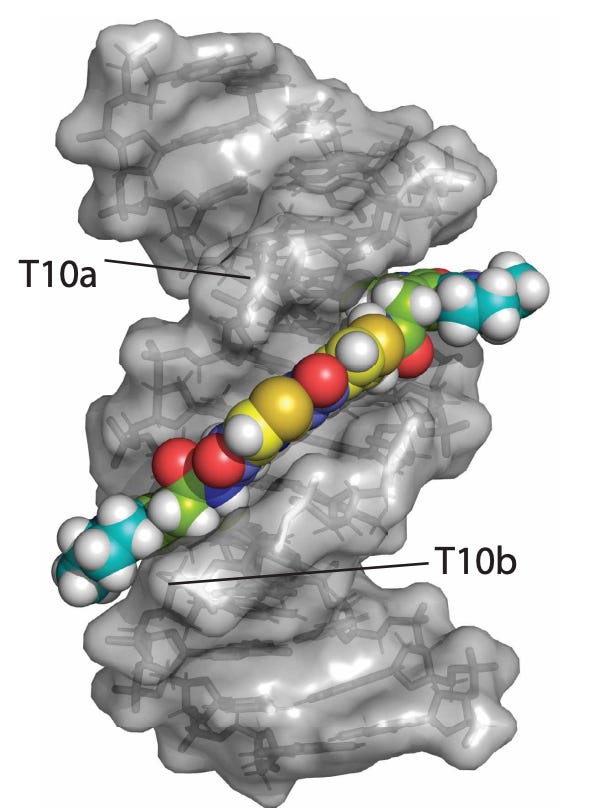
As it turns out, this structure slots very nicely into DNA. The bond ends up looking like this:
The mechanism for how this molecule causes damage is simple but beautiful. It slides into the minor groove of the DNA helix, while barely able to hold its unstable warhead together. Once it is settled, the strained chemical bonds on colibactin snap and rapidly react with the closest atoms in the vicinity - in this case, those on the DNA. The resulting bond is permanent.
The type of damage this causes is extremely hard for a cell to fix. It does not look anything like the type of damage cells are used to experiencing. The damage that colibactin induces is the equivalent of supergluing together two complementary strands of DNA. The cell is not able to use the DNA for anything - transcription or replication - because that requires the cell to unwind the DNA strands together, which it now cannot do. The best the cell can do is either ignore it, or cut out the region and copy in the correct sequence from the other chromosome (which is hopefully undamaged!). This type of repair has a high probability of inducing a mutation.
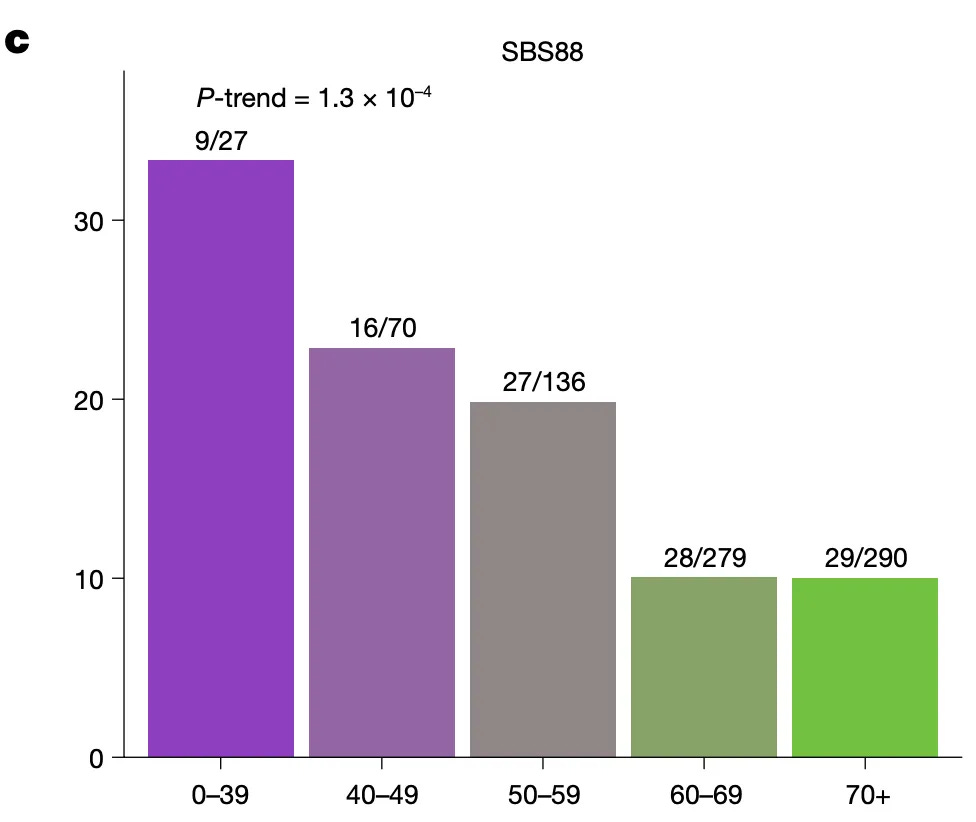
How does this tie back to early-onset colorectal cancer? It turns out the type of DNA damage that colibactin induces leaves a particular mutational signature - a DNA scar, if you will. And this signature (labeled SBS88) is highly enriched in colorectal cancers from young patients.
So what can we do about it?
Option 1: Reduce the amount of colibactin-producing bacteria
If the core problem is that we have problematic toxin-producing bacteria in the gut, then the most straightforward approach would be to eliminate them. Unfortunately, this is tricky. Antibiotics are not selective, and some of them can increase the proportions of pathogenic bacteria. On the flip side, drugs that down-regulate the immune system can be paradoxically helpful - E. coli thrive on oxygen-rich environments that immune cells bring - but come with tradeoffs. Probiotics are promising, as introducing symbiotic and commensal bacteria inherently decreases the proportion of problematic pks+ E. coli, but the only study to try this in an animal model introduced, ironically, another bacteria that produces colibactin3. The most promising study to date used putrescine in supplement fashion to decrease the growth of pks+ E. coli, with the general idea of inhibiting the growth of toxin-producing bacteria. In general, the idea of modulating the microbiome has a great deal of promise, but has struggled immensely in the clinic4.
Option 2: Detoxify colibactin
You may be wondering how pks+ E. coli themselves do not fall victim to colibactin, as, after all, they also have DNA that is vulnerable to damage. The answer is that they have evolved their own antidote: an enzyme that breaks the DNA-stapling warhead.
Could we create something similar? Or otherwise create something that would prevent bacteria from producing colibactin in the first place?
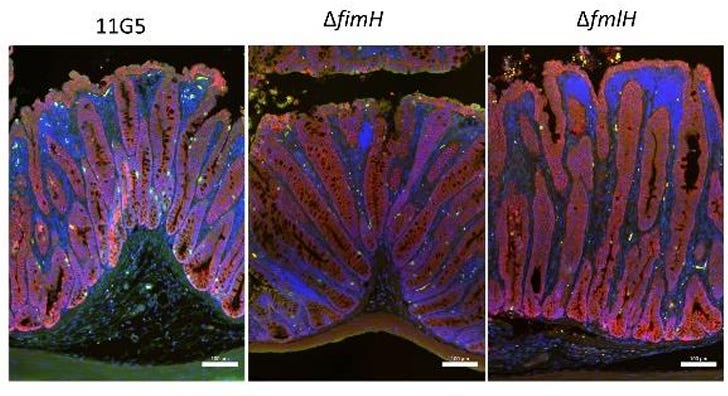
The answer is yes! And many of the techniques that researchers have used are quite clever. A group in the Netherlands showed that colibactin-producing bacteria need to stick to epithelial cells in the gut to cause harm. If you block the ability to stick by either knocking out a gene (see figure below), or by adding a drug, the amount of damage is dramatically reduced.
Another study used a boron-containing drug to mimic one of the substrates that bacteria use to create colibactin, which led to a large drop in production. A third paper directly took advantage of the pks+ E. coli detoxifying enzyme by fusing it to the cell surface of a different bacterial strain, generating the biological equivalent of a gut colibactin vacuum cleaner. If you could co-colonize your microbiome with bacteria that break down any colibactin outside of a cell, that should reduce the total gut toxin load significantly. It also points towards a synthetic biology utopian future where one could engineer microbes to detoxify many different toxins. It’s an exciting world of possibilities.
Option 3: Protect DNA directly
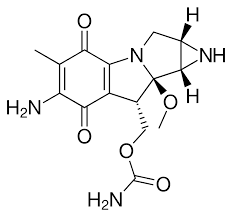
Colibactin is a very unique mutagen, but it does have some chemical cousins. One of the closest is mitomycin C, a chemotherapy drug. It looks like this:
It lacks the flat structural piece to bind into the minor groove, but it does resemble guanosine enough to associate with DNA. This, just like with colibactin, leads to the activation of the warhead (same triangular piece, though with a nitrogen this time around) with irreversible crosslinking of DNA. It works as a chemotherapy due to the high division rate in cancerous cells, though it comes with (predictable) negative side effects.
Interestingly, there are chemical compounds that may be able to block damage induced by crosslinking agents. A 2008 study tested administering amifostine prior to mitomycin C exposure, and showed a reduction of DNA damage back to baseline:
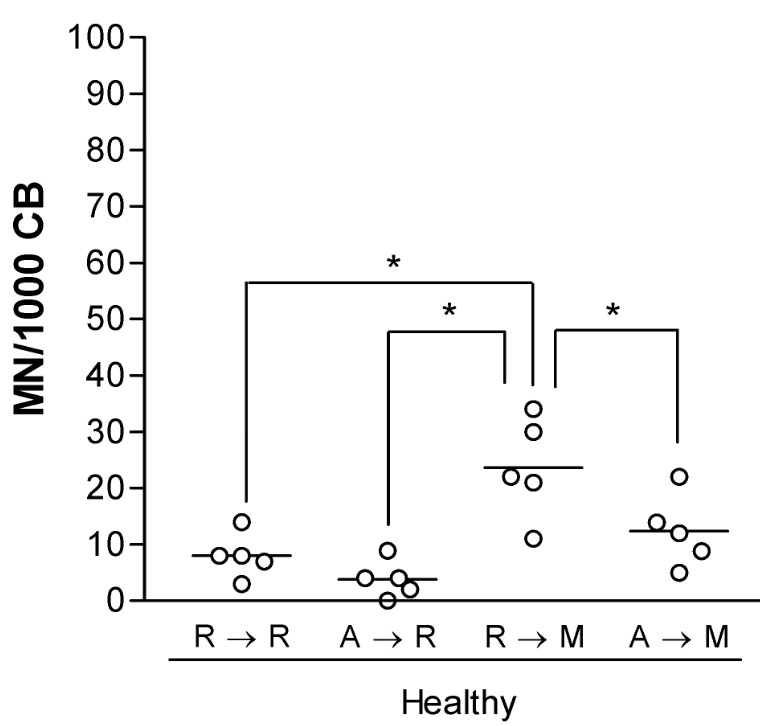
The impetus for this study came from wanting to find ways to alleviate DNA damage in patients with Fanconi anemia, a rare disease where the genes that can repair interstrand crosslinks are mutated. If you can protect DNA from agents like mitomycin C, the implications extend well beyond colibactin-producing bacteria.
So where are we now?
Oftentimes, epidemiological problems feel intractable to scientific solutions. Solving the primary driver of lung cancer (smoking) did not come from new drugs that broke the toxins in tobacco, but rather from public-focused anti-smoking campaigns.
In this case, science may actually have the solution. We know the driver of a large fraction of early-onset colorectal cancer cases, and we have scalable ways of alleviating it. We may be early (you’ll note nearly every paper I cited in this piece was released within the last three years), but the proposed interventions do not seem particularly sci-fi. Probiotic solutions, in particular, could move very quickly from being a proof-of-concept to something sitting on a store shelf (or a fridge, in this case).
The other big takeaway is in seeing the medical problem for its molecular drivers. Cardiovascular disease, like early-onset colorectal cancer, is affected by high-level lifestyle components like diet and exercise. And yet, understanding the importance of cholesterol led to the life-saving drug category of statins.
We should be able to intervene in the causes of cancer in a similar way.
Albeit with the caveat that colorectal cancer in children is rare, totaling ~ 100 total cases in the United States per year.
We largely do not have deep profiling of microbiomes and cancers from individuals diagnosed in the 1990s that we can compare to.
Clinical trials to solve this problem seem to be underway.
A complex topic worthy of a much longer, separate post.


Awesome overview! Super interesting stuff. Could colibactin exposure increase the risk of any other cancers, or is it just linked to colorectal?
Fantastic writeup, thanks for putting this together!
And now I'm going to do that annoying thing and ask... what do you think is the primary driver or increased levels of colibactin in the gut? Seems pretty clearly environmental, so what might young people be consuming that's skewing their gut bacteria in a harmful manner?